|
Wood was the original choice but it deteriorated in the electrolyte. The gases created during charge are absorbed and there is no water loss if venting can be prevented.Įarly separators were made of rubber, glass fiber mat, cellulose and polyethylene plastic. The tightly wound or stacked separator/electrode arrangement forms a solid mechanical unit that offers similar performance to the flooded type but is smaller and can be installed in any orientation without leakage. With the development of the sealed nickel-cadmium in 1947 and the maintenance-free lead acid in the 1970s, the electrolyte is absorbed into a porous separator that is compressed against the electrodes to achieve chemical reaction. Ion flow through the separator of Li-ion īattery separators provide a barrier between the anode (negative) and the cathode (positive) while enabling the exchange of lithium ions from one side to the other.Įarly batteries were flooded, including lead acid and nickel-cadmium. Figure 1 illustrates the building block of a lithium-ion cell with the separator and ion flow between the electrodes.įigure 1. Self-discharge eventually depletes the charge of a battery during prolonged storage. The small amount of current that may pass through the separator is self-discharge and this is present in all batteries to varying degrees. Although ions pass freely between the electrodes, the separator is an isolator with no electrical conductivity. Ions are atoms that have lost or gained electrons and have become electrically charged. The separator is moistened with electrolyte and forms a catalyst that promotes the movement of ions from cathode to anode on charge and in reverse on discharge. Note: If the Copper electrode is not pure Cu, an alloy, perhaps containing noble Silicon, you may be performing an ascribed path to H2SO4 preparation (see, for example, ), so they will be a definite rise in pH along with problematic explosive hydrogen generation.The building blocks of a battery are the cathode and anode, and these two electrodes are isolated by a separator. 
Where the above theory corresponds to what is observed, per my experience, the copper anode has been cleaned and also the cathode possibly from increased acidity. To give a different example, here is a half-reaction involving lead: In the half-reaction in question, copper changes oxidation states, and the copper ions balance out the charge of the electrons so that both sides of the half-reaction have equal charge (zero, in this case). The species that balance the charges, however, might or might not undergo a change in oxidation state. Oxidation state imbalance of half-reactionsĪt least one atom (as element, simple ion, or part of a compound or complex ion) has to undergo a change in oxidation state. The ions (in solution) are also responsible for the charge transport between anode and cathode. In a oxidation, we produce electrons, so either we need to produce cations at the same time or consume anions (or have ions on both sides of the equation, but of unequal charge). 
So if electrons appear in the half-reaction, you need some ions to balance it out. There is no physical state associated with the electrons, but they are removed through the wire. For the cell in question, the oxidation half-reaction at the anode is: 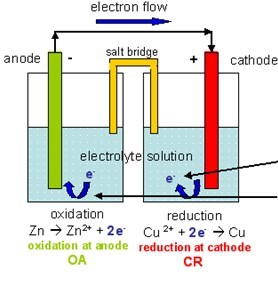
Half-reactions have electrons either as reactants (reduction half-reaction at the cathode) or as products (oxidation half-reaction at the anode). 
This is one way of achieving charge balance, but there are many other ways. I've been given to understand that in electrolysis, the negative ions move to the anode, and lose electrons to the anode.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
